7.0 DEFINITION OF SOLUBILITY
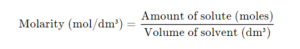
The term solubility refers to the maximum amount of solute that will dissolve in a given amount of solvent at a given temperature to produce a stable solution.
In terms of mole per dm3, solubility is the maximum amount of solute in moles or grams that will saturate 1dm3 of a solvent, at a particular temperature.
7.1 SOLUTION AND ITS TYPES
A solution is a uniform or homogeneous mixture of two or more substances. An aqueous solution is a solution with water as its solvent.
- SATURATED SOLUTION – A solution that contains the maximum amount of solute that can dissolve at a specific temperature. Any additional solute will not dissolve and may form a precipitate.
- UNSATURATED SOLUTION – A solution that contains less solute than it can hold at a given temperature. More solute can be dissolved without changing the temperature until the solution becomes saturated.
- SUPERSATURATED SOLUTION – A solution is one which contains more of the solute than it can normally hold at that temperature. This can be achieved by heating the solution to dissolve more solute and then cooling it gently without allowing the excess solute to crystallize.
7.2 SOLUBILITY CURVE
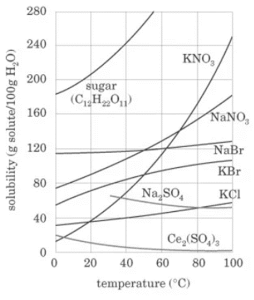
A solubility curve shows the relationship between temperature and the amount of solute that can dissolve.
You can deduce from the curve how much solute will dissolve at a specific temperature or whether a solution is unsaturated, saturated, or supersaturated.
As temperature increases, solubility of solids generally increases (more solute can dissolve), while the solubility of gases decreases i.e Solubility is Temperature-Dependent.
SOLUTE – A dissolved substance, which may be solid, liquid or gas.
7.3 SOLVENT
A solvent is a substance that can dissolve a solute, forming a homogeneous solution. The most known example of solvent is water; others include ethanol, methanol, acetone, benzene etc. Solvents could be solid, liquid or gaseous (usually liquid).
Different solvents are used to dissolve fats, oils, and paints because these substances are non-polar, meaning water (a polar solvent) is ineffective at dissolving them. Common solvents include:
- Fats and oils: Dissolved by organic solvents like alcohol, petrol, kerosene, benzene, ether, and acetone.
- Paints: Solvents like turpentine and white spirit are used for thinning or removing paints.
NATURE OF SOLVENT – Water and other polar solvents dissolve most inorganic, ionic and polar substances and few organic ones. On the other hand, organic/non-polar solvents like ethanol, acetone, turpentine etc. are usually used to dissolve organic substances.
USES OF SOLVENT
- Solvents are used as cleaning agents e.g petrol, acetone, alcohol, spirits etc.
- They are used as paint thinners and varnishers e.g turpentine.
- They are used in the pharmaceutical industries to dissolve active ingredients for easy absorption by the body.
- It provides a medium for chemical reactions in laboratories/industries.
- Used in glue and adhesive formulations.
- They are used in the production of perfumes to dissolve essential oils and blend fragrances e.g ethanol.
7.4 TRUE AND FALSE SOLUTIONS
TRUE SOLUTIONS – A true solution is a homogeneous mixture where solute is completely dissolved. Examples: an aqueous solution of sodium chloride and copper (II) tetraoxosulphate (VI).
FALSE SOLUTIONS – A false solution is a mixture where the solute particles are not fully dissolved in the solvent, resulting in a system that appears homogeneous but is actually heterogeneous. False solutions include suspensionsand colloids, where particles are larger than in true solutions.
- Suspensions – A heterogeneous mixture where solid particles are dispersed in a liquid or gas but are large enough to settle out if left undisturbed. Examples: Harmattan haze, water-based paints.
- Colloids – A mixture where fine particles are dispersed evenly throughout another substance, but the particles are small enough to remain suspended and not settle out. Examples: Fog (liquid in gas), milk (liquid in liquid), aerosol sprays (liquid in gas), rubber solution, emulsion paints.
DTW Tutorials Study Resource Links;
First of All to obtain high JAMB &WAEC Scores, YOU HAVE TO Practice! Practice!! Practice!!
Use DTW JAMB & WAEC 2025 CBT Practice App!!!
– GET DTW TUTORIALS JAMB & WAEC 2025 CBT EXAM PRACTICE APP for all Subjects with over 31,000 Past Questions and Correct Solutions to Practice with offline! (Activation cost is N4000 for 1 year) Download Links Below for Mobile Phones & Laptop Computer;
DTW TUTORIALS JAMB 2025 APP For MOBILE Phone Direct Download link;
https://play.google.com/store/apps/details?id=com.iafsawii.dtw.jamb
DTW TUTORIALS JAMB 2025 APP For DESKTOP Laptop Computer Direct Download link; https://drive.google.com/file/d/1iIHBoWjEeJeCFyTO9nt-9kAveH2FqjrT/view?usp=sharing
Download Links for WAEC 2025 App;
JAMB RESOURCE LINKS BELOW;
– JAMB Past Questions Solved Playlists on Math, Phy, Chem; https://www.youtube.com/playlist?list=PLLgYU6fS5143-p4dfWIFL7keuB1SBgT2b
– THE LEKKI HEADMASTER – Summary, Questions And Answers (JAMB 2025 NOVEL); https://dtwtutorials.com/the-lekki-headmaster-jamb-2025-novel-summary-questions-and-answers-pdf-download/
– JAMB 2025 Recommended Text Books – https://dtwtutorials.com/jamb-2025-recommended-text-books-for-all-subjects/
– JAMB 2025 Syllabus all Subjects – https://dtwtutorials.com/jamb-2025-syllabus-free-download/
– JAMB 2025 Syllabus in 30 Days Timetable Challenge by DTW Tutorials for Science, Art & Commercial Subject Combinations – Cover Your JAMB Syllabus in 30 Days Challenge; https://dtwtutorials.com/jamb-2025-syllabus-in-30-days-timetable-challenge-by-dtw-tutorials-cover-your-jamb-syllabus-in-30-days-challenge/
– How to Manage Your Jamb Exam Time for High Scores; https://youtu.be/Tp4Va8haib8
– Physics Notes and Questions on All topics; https://dtwtutorials.com/category/tutorials/physics-tutorials/
– Chemistry Notes and Questions on All topics; https://dtwtutorials.com/category/tutorials/chemistry/
– How to Read, Understand and Remember Always- https://youtu.be/kL8BpRePudA
– How to Cover Your JAMB Syllabus Fast in 30 Days!!; https://youtu.be/RVgyn01Ptd0
– What to do a night before your Jamb Exam (+Exam Prayers); https://youtu.be/njbAx4Oz5Rw
– How to Manage Your Jamb Exam Time for High Scores; https://youtu.be/Tp4Va8haib8
– Overcoming Exam Fear/Anxiety– https://youtu.be/Uvf81rvd0ls
You can also join our online groups below for instant JAMB 2025 Updates;
Join DTW JAMB 2025 Intensive Tutorials Study Groups on Facebook, Telegram and WhatsApp Group;
Facebook Group – https://web.facebook.com/groups/dtwtutorialsgroup/
WhatsApp Group – https://chat.whatsapp.com/E8pprCQYtahKfpQN9UB0aU
Telegram Group – https://t.me/+AcXfhJPSIiI2ZTY0
WhatsApp Channel – https://whatsapp.com/channel/0029VaAWvTmDDmFT9o25dV3u
DTW JAMB 2025 Intensive Online Lessons/Tutorials
Online JAMB 2025 Tutorials – Your Path to Jamb Success!
Are you preparing for the JAMB 2025 Exam and aiming for excellence? Look no further than Online Jamb Tutorial by DTW Consult. We’re dedicated to helping you ace your Jamb with confidence.
- Why Choose DTW Online JAMB Intensive Tutorials?
• Engaging, Clear and Interactive Online Lectures
• Completion of JAMB Syllabus
• Weekly Quiz Assessments
• Continuous Brainstorming and Competitions
• Membership in an Active Learning Community
• Consistent Solving of JAMB Past Questions-
• Expert Jamb Instructors
• Comprehensive Study Materials - All Classes are Recorded!! In case you miss any class, and when you join us you will have access to all the previous class recorded videos!!!
• Subjects;
English
Physics
Chemistry
Biology
Math
Economics
Literature
Crs
Government
• Affordable Tuition – N7000 monthly (6pm – 10pm, Mon to Fri)
Lectures Ongoing! Register Now!!
Bank Details:
Account Name: DTW Consult
Account Number: 6414330770
Bank: Moniepoint
Amount – N7000
For easy payment and enrollment.
Proof of payment should be sent by WhatsApp.
Contact Us:
WhatsApp: 09085099582, 08038732879
Email: dtwconsultng@gmail.com
Take a step closer to your Jamb success with DTW Online Jamb 2025 Intensive Tutorials.
Let’s work together to unlock your full potential!

https://youtu.be/P7wtBH46ZMMnsive Tutorials. Let’s work together to unlock your full potential! #JambPrep #OnlineTutorial #DTWConsult #JambSuccess #jamb2025 #utme2025

No Comments