13.0 MEANING OF RATE OF REACTION
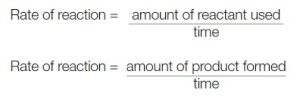
The rate of chemical reaction can be defined as the number of moles of reactant converted or product formed per unit time. Reaction rates are found by measuring the rate at which the mass or concentration of a:
- Reactant is decreasing.
- Product is increasing.
13.1 FACTORS AFFECTING THE RATE OF REACTION
- TEMPERATURE – Reactions are faster at higher temperatures and slower at lower temperatures. Increasing temperature gives particles more kinetic energy, leading to more frequent and more energetic collisions, thus increasing the rate of the reaction.
CONCENTRATION/PRESSURE – In solutions, an increase in concentration of reactants leads to an increase in the rate of reaction and vice versa. For example, the rate of the reaction between HCl and Na₂S₂O₃ increases as the concentration of HCl increases.
For gases, increasing pressure reduces the volume, increasing the number of collisions and thus increasing the reaction rate. For example, the effect of pressure on reactions involving gases such as in the Haber process:
(N₂ + 3H₂ → 2NH₃).
- SURFACE AREA – When a solid reactant is broken into smaller pieces or powdered, its surface area increases, exposing more particles to collisions. For example, the reaction between marble (CaCO₃) and HCl, comparing the powdered form vs. lumps of the same mass. Powdered substances have a larger surface area than lumps, leading to more frequent collisions at the surface and thus a faster reaction. Therefore, an increase in surface area increases the rate of reaction; while a decrease in surface area decreases the rate of reaction.
CATALYST – A catalyst is a substance which alters the rate of a chemical reaction and remains the same at the end of the reaction (it doesn’t take part in the reaction). The presence of a catalyst provides an alternative reaction pathway with lower the activation energy, increasing reaction rates without being consumed in the reaction. For example, in the decomposition of hydrogen peroxide (H₂O₂) or potassium chlorate (KClO₃), the presence of manganese dioxide (MnO₂) – as a catalyst speeds up the reaction process.
SOME REACTIONS AND THEIR CATALYST
NATURE OF REACTANTS – The chemical nature of reactants determines the rate of reaction as different substances have different energy contents. Some substances react faster than others based on their molecular structure and bond strength.
- LIGHT – Some reactions are influenced by light. For example, the reaction between hydrogen and chlorine. Such reactions are known as photochemical reactions. In these reactions, the reactant molecule becomes activated on absorbing light energy, and reacts rapidly together in a series of chain reactions.
It is important to note that the factors which affects the (a) energy content of reacting particles (b) activation energy of the reaction (c) frequency of effective collision; will also affect the rate of reaction.
13.2 RELATIONSHIP BETWEEN KINETIC THEORY AND RATE OF REACTION
The kinetic theory states that particles are constantly moving and colliding. Reaction rates depend on the frequency and energy of these collisions, which are influenced by factors such as temperature, concentration, and surface area.
COLLISION THEORY – It states that for a chemical reaction to occur, the reacting particles must collide with sufficient energy (equal to or greater than the activation energy) and in the correct orientation. It is a principle of chemistry used to predict the rates of chemical reactions.
ACTIVATION ENERGY – It is defined as the minimum amount of energy which reactant particles must possess for a chemical reaction to occur (i.e. reactants to form products). A higher activation energy means a slower reaction, as fewer particles have enough energy to overcome this barrier. Lowering the activation energy (e.g., using a catalyst) speeds up the reaction.
ARRHENIUS LAW/EQUATION – The Arrhenius law states that the motion of the structural units (atoms, molecules, ions, etc.) should slow down at a slower rate through the glass transition than is experimentally observed. The equation is commonly given in the form of an exponential function; in simpler words, it predicts that a small increase in reaction temperature will produce a marked increase in the magnitude of the reaction-rate constant. The rate of a reaction depends on the temperature and the activation energy (Ea).
DTW Tutorials Study Resource Links;
First of All to obtain high JAMB &WAEC Scores, YOU HAVE TO Practice! Practice!! Practice!!
Use DTW JAMB & WAEC 2025 CBT Practice App!!!
– GET DTW TUTORIALS JAMB & WAEC 2025 CBT EXAM PRACTICE APP for all Subjects with over 31,000 Past Questions and Correct Solutions to Practice with offline! (Activation cost is N4000 for 1 year) Download Links Below for Mobile Phones & Laptop Computer;
DTW TUTORIALS JAMB 2025 APP For MOBILE Phone Direct Download link;
https://play.google.com/store/apps/details?id=com.iafsawii.dtw.jamb
DTW TUTORIALS JAMB 2025 APP For DESKTOP Laptop Computer Direct Download link; https://drive.google.com/file/d/1iIHBoWjEeJeCFyTO9nt-9kAveH2FqjrT/view?usp=sharing
Download Links for WAEC 2025 App;
JAMB RESOURCE LINKS BELOW;
– JAMB Past Questions Solved Playlists on Math, Phy, Chem; https://www.youtube.com/playlist?list=PLLgYU6fS5143-p4dfWIFL7keuB1SBgT2b
– THE LEKKI HEADMASTER – Summary, Questions And Answers (JAMB 2025 NOVEL); https://dtwtutorials.com/the-lekki-headmaster-jamb-2025-novel-summary-questions-and-answers-pdf-download/
– JAMB 2025 Recommended Text Books – https://dtwtutorials.com/jamb-2025-recommended-text-books-for-all-subjects/
– JAMB 2025 Syllabus all Subjects – https://dtwtutorials.com/jamb-2025-syllabus-free-download/
– JAMB 2025 Syllabus in 30 Days Timetable Challenge by DTW Tutorials for Science, Art & Commercial Subject Combinations – Cover Your JAMB Syllabus in 30 Days Challenge; https://dtwtutorials.com/jamb-2025-syllabus-in-30-days-timetable-challenge-by-dtw-tutorials-cover-your-jamb-syllabus-in-30-days-challenge/
– How to Manage Your Jamb Exam Time for High Scores; https://youtu.be/Tp4Va8haib8
– Physics Notes and Questions on All topics; https://dtwtutorials.com/category/tutorials/physics-tutorials/
– Chemistry Notes and Questions on All topics; https://dtwtutorials.com/category/tutorials/chemistry/
– How to Read, Understand and Remember Always- https://youtu.be/kL8BpRePudA
– How to Cover Your JAMB Syllabus Fast in 30 Days!!; https://youtu.be/RVgyn01Ptd0
– What to do a night before your Jamb Exam (+Exam Prayers); https://youtu.be/njbAx4Oz5Rw
– How to Manage Your Jamb Exam Time for High Scores; https://youtu.be/Tp4Va8haib8
– Overcoming Exam Fear/Anxiety– https://youtu.be/Uvf81rvd0ls
You can also join our online groups below for instant JAMB 2025 Updates;
Join DTW JAMB 2025 Intensive Tutorials Study Groups on Facebook, Telegram and WhatsApp Group;
Facebook Group – https://web.facebook.com/groups/dtwtutorialsgroup/
WhatsApp Group – https://chat.whatsapp.com/E8pprCQYtahKfpQN9UB0aU
Telegram Group – https://t.me/+AcXfhJPSIiI2ZTY0
WhatsApp Channel – https://whatsapp.com/channel/0029VaAWvTmDDmFT9o25dV3u
DTW JAMB 2025 Intensive Online Lessons/Tutorials
Online JAMB 2025 Tutorials – Your Path to Jamb Success!
Are you preparing for the JAMB 2025 Exam and aiming for excellence? Look no further than Online Jamb Tutorial by DTW Consult. We’re dedicated to helping you ace your Jamb with confidence.
- Why Choose DTW Online JAMB Intensive Tutorials?
• Engaging, Clear and Interactive Online Lectures
• Completion of JAMB Syllabus
• Weekly Quiz Assessments
• Continuous Brainstorming and Competitions
• Membership in an Active Learning Community
• Consistent Solving of JAMB Past Questions-
• Expert Jamb Instructors
• Comprehensive Study Materials - All Classes are Recorded!! In case you miss any class, and when you join us you will have access to all the previous class recorded videos!!!
• Subjects;
English
Physics
Chemistry
Biology
Math
Economics
Literature
Crs
Government
• Affordable Tuition – N7000 monthly (6pm – 10pm, Mon to Fri)
Lectures Ongoing! Register Now!!
Bank Details:
Account Name: DTW Consult
Account Number: 6414330770
Bank: Moniepoint
Amount – N7000
For easy payment and enrollment.
Proof of payment should be sent by WhatsApp.
Contact Us:
WhatsApp: 09085099582, 08038732879
Email: dtwconsultng@gmail.com
Take a step closer to your Jamb success with DTW Online Jamb 2025 Intensive Tutorials.
Let’s work together to unlock your full potential!

https://youtu.be/P7wtBH46ZMMnsive Tutorials. Let’s work together to unlock your full potential! #JambPrep #OnlineTutorial #DTWConsult #JambSuccess #jamb2025 #utme2025

No Comments