ATOMIC STRUCTURE
The structure of an atom consists of three main components: protons, neutrons, and electrons, arranged in the following order:
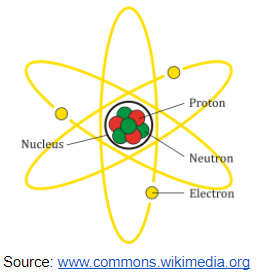
- Nucleus – The nucleus is the atom’s central, dense core. It contains two types of subatomic particles: Proton (positively charged), and Neutron (neutral – no charge).
- Electrons – They are negatively charged particles that move in regions of space around the nucleus called electron shells or orbitals. Electrons are responsible for the atom’s chemical properties and bonding.
- WORKS OF PIONEERS IN ATOMIC THEORY
John Dalton (1766-1844) – In the year 1803, he proposed a theory called Dalton’s Atomic Theory. It states that:
ü All elements are made up of small, indivisible particles called atoms.
ü Atoms can neither be created nor destroyed.
ü Atoms of the same element are alike in every aspect and differs from atoms of other elements.
ü Atoms of different elements combine in simple ratios to form compounds.
ü All chemical changes result from the combination or separation of atoms.
J.J. Thomson (1856-1940)
ü Discovery of the Electron (1897) – Thomson’s experiments with cathode rays led to the discovery of the electron, the first subatomic particle. He demonstrated that these rays were streams of negatively charged particles (electrons). He proposed the plum pudding model, suggesting that atoms were made of a positively charged sphere with negatively charged electrons embedded within it.
Henry Moseley (1887-1915)
ü Moseley’s Law (1913) – Moseley studied the X-ray spectra of elements and discovered that the frequency of X-rays emitted by each element was related to the element’s atomic number (the number of protons). This demonstrated that atomic number, not atomic mass, determines the identity of an element.
Robert Millikan (1868-1953)
ü Millikan’s Oil Drop Experiment (1909) – Millikan measured the charge of the electron through his famous oil drop experiment. By suspending tiny, charged oil droplets between two electric plates and adjusting the voltage, he was able to determine the charge of an electron as approximately 1.6×10−19 coulombs.
Ernest Rutherford (1871-1937)
ü Rutherford’s Gold Foil Experiment (1909) – Rutherford conducted an experiment where alpha particles were directed at a thin gold foil. While most particles passed through the foil, some were deflected at sharp angles, suggesting the presence of a dense, positively charged center in the atom. This led to the development of the nuclear model of the atom, which proposed that the atom consists of a small, dense nucleus containing protons, with electrons orbiting around it.
Niels Bohr (1885-1962)
ü Bohr’s Atomic Model (1913) – Bohr expanded upon Rutherford’s nuclear model by introducing quantum theory to explain the behavior of electrons. He proposed that electrons orbit the nucleus in specific, quantized energy levels, and that electrons could jump between these levels by absorbing or emitting energy in discrete amounts (quanta). This model explained the discrete lines in the hydrogen emission spectrum.
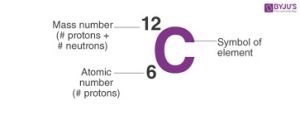
ATOMIC NUMBER – This is the total number of proton(s) in the nucleus of an atom of an element. It is represented by the letter Z.
MASS NUMBER – This is the sum of protons and neutrons in the nucleus of an atom of an element, denoted by A.
ELECTRONIC CONFIGURATION – This is the arrangement of electrons in the shell or orbital of an atom of an element.
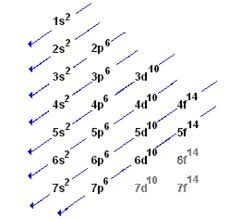
- SHELL ELECTRONIC CONFIURATION – Electrons in shells nearer the nucleus possess less energy than those farther away. Starting from the shell nearest the nucleus, the shells are named as K, L, M, N, …, these corresponds to the energy levels numbered 1, 2, 3, 4, …, The maximum number of electrons in a shell is given by the formula 2n2, where n is the energy level of the shell. Electrons are filled in the K-shell (n=1) first, followed by L, M, and N-shells with respective limits.
K = 2(1)2 = 2 (where n is 1)
L = 2(2)2 = 8 (where n is 2) etc.
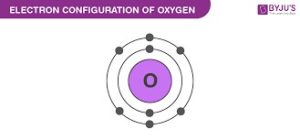
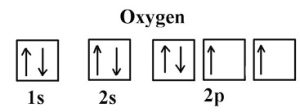
ORBITAL ELECTRONIC CONFIGURATION – the arrangement of electrons in orbitals around an atomic nucleus. For example, oxygen; 8 electrons has its orbital configuration as shown on the diagram.
- THE FOUR QUANTUM NUMBERS
- PRINCIPAL QUANTUM NUMBER (n) has an integral value 1, 2, 3, 4, etc. the energy levels or shell are determined by the principal quantum number.
- SUBSIDIARY / AZUMUTUAL QUANTUM NUMBER (l) has integral values ranging from 0 to (n -1). The electrons with subsidiary quantum numbers 0, 1, 2 and 3 are usually referred to as the s, p, d, and f electrons respectively. Thus, this quantum number shows how many energy sublevels there are in each electron.
- MAGNETIC QUANTUM NUMBER (m) which has integral values ranging from-1 through 0 to 1, shows the number of orbitals in each energy sublevel e.g. the p subshell has three p orbitals.
- SPIN QUANTUM NUMBER (s), has values -1/2 and +1/2. An orbital can only hold two electrons which spin on their axes in opposite directions.
- PRINCIPLES THAT GOVERN ELECTRON FILLING INTO ORBITALS
- PAULI EXCLUSION PRIINCIPLE – This principle states that the same orbital of an atom cannot have the same values for all the four quantum numbers. For example, two electrons with n = 1, l = 0, m = o will differ in s, because one will be s =-1/2.
- HUND’S PRINCIPLE – This rule states that electrons occupy each orbital singly first before pairing takes place in a degenerate orbital.
- AUFBAU PRINCIPLE – This principle states that in building up an atom, electrons are filled into orbitals in order of increasing energy. i.e. electrons are fed into orbitals starting at the lowest energy level before filling higher energy levels.
- SHAPES OF ORBITALS
- S ORBITAL – Spherical in shape.
- P ORBITAL – Dumbbell in shape.
- D ORBITAL – Double dumbbell in shape.
- F ORBITAL – Fundamental in shape.
- ISOTOPES
These are atoms of a particular element with the same atomic number but different mass number, due to the difference in the number of neutrons present in these atoms. The phenomenon resulting to isotopes is called isotopy. Examples of isotopes include 3517 Cl and 3717 Cl; 11 H, 21 H and 31 H etc.
- CHEMICAL COMBINATON
This is the formation of a chemical bond between two or more atoms, molecules or ions to give rise to a stable chemical compound. Therefore, to attain chemical stability, chemical combination must occur.
TYPES OF CHEMICAL COMBINATION
- Electrovalent (or ionic) combination
- Covalent combination
- Ordinary covalent combination
- Co-ordinate (or dative) covalent combination.
- There is also metallic combination.
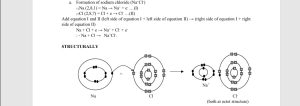
ELECTROVALENT COMBINATION – This is a bonding where there is transfer of electrons from one atom (usually metallic) to another (usually non-metallic). the electrons involved resides in the outermost shell of the atoms and are known as valence electrons. Example:
CHARACTERISTICS OF ELECTROVALENT COMPOUNDS
- They are formed out of electrovalent combination.
- They exist mainly as solids at room temperatures.
- They have high melting points and boiling points because of the strong ionic bonds between the ions.
- Most of them readily dissolve in water, but some do not like MgO do not dissolve in water.
- In their liquid state they are electrolyte because they conduct electricity, due to the free movement of the ions.
COVALENT COMBINATION
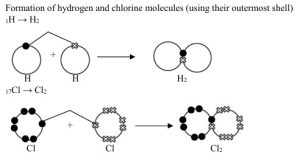
Ordinary Covalent Combination – In this combination there is sharing of electrons between the reacting atoms so that both attain noble gas configurations. For example,
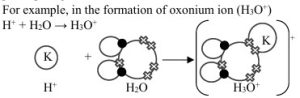
Co-Ordinate Covalent Combination – This is also known as dative covalent combination. Like the ordinary covalent combination, there is sharing of electrons, but unlike the ordinary covalent combination the electrons shared in the dative covalent combination is donated by one of the participating atoms.
CHARACTERISTICS OF ORDINARY COVALENT COMPOUND
- They have definite shape. Simple covalent compounds are often either gases or volatile liquids.
- They usually dissolve in non-polar solvents like ethanol and not polar solvent like water.
- They are mostly non-electrolytes.
- OTHER TYPES OF BONDING FORCES
- Van Der Waal’s Force – These are short-range attractive forces operating between discrete molecules. They operate only when the particles are in close contact with one another as in liquids and solids.
- Hydrogen Bond – This arise when hydrogen is covalently linked to elements like N, O, and F. they tend to pull the shared pair of electrons in the covalent bonds towards themselves, resulting in the formation of dipole where the hydrogen atom is partially positive, while the N, O or F atom is partially negative.
- Metallic Bonds – This is the force of attraction which holds metal atoms together in crystal lattice.
- SHAPES OF MOLECULES
Shapes of molecules are defined by the spatial arrangement of atoms, influenced by bonding and lone electron pairs’ repulsion. The shapes of simple molecules are determined by their molecular geometry, which depends on the number of bonding pairs and lone pairs of electrons around the central atom.
TYPES OF SHAPES OF MOLECULES
- Linear – Atoms combine to form a straight line, making it a linear molecule e.g H₂ (Hydrogen gas), O₂ (Oxygen gas), Cl₂ (Chlorine gas), HCl (Hydrogen chloride), CO₂ (Carbon dioxide) etc. It has a bond angle of 180o.
- Bent or V-shaped – Formed by a central atom and another atom with two lone pairs of electrons, that repels the bonding pairs e.g H₂O (Water). It has a bond angle of about 104.5o.
- Tetrahedral – Formed in a molecule with central atom and four other atoms, bonded symmetrically in a tetrahedral shape e.g CH₄ (Methane). It has a bond angle of about 107.5o.
- Pyramidal – Formed in a molecule with central atom and three other atoms, bounded in a pyramidal shape e.g NH₃ (Ammonia). It has a bond angle of about 109o.
- THE PERIODIC TABLE
The most important classification in chemistry is the arrangement of the element in the periodic table. This was first done by a Russian scientist, Dmitri Mendeleev in 1869. The periodic table is an ordered arrangement of the chemical elements into rows (periods) and columns (groups) in accordance with their increasing atomic number and recurring chemical properties.
THE MODERN PERIODIC LAW – It states that the physical and chemical properties of an element are the periodic function of their atomic numbers.
GROUPS – These are the vertical columns in the periodic table, numbered from 0 to 7 (i.e 8 groups). Elements in the same group have the same number of valence electrons.
PERIODS – These are the horizontal rows in the periodic table, numbered from 1 to 7(i.e 7 periods). Elements in the same period have the same number of electron shells.
- PROPERTIES VARIATION IN THE PERIODIC TABLE
- Atomic radius – Increases down the group, decreases across the period.
- Ionic radius – Increases down the group, decreases across the period.
- Electronegativity – Decreases down the group, increases across the period.
- Electron affinity – Decreases down the group, increases across the period.
- Ionization energy – Decreases down the group, increases across the period.
Group 1 – Alkaline metals Group 5 – Nitrogen family (Pnictogens).
Group 2 – Alkaline earth metals. Group 6 – Oxygen family (Chalcogens).
Group 3 – Boron family (Icosagens). Group 7 – Halogens.
Group 4 – Carbon family (Tetragens). Group 0/8 – Noble gases.
TRANSITION METALS – These are elements between group 2 and 3 of the periodic table.
LANTHANIDES AND ACTINIDES – These are typically found in separate rows at the bottom of the periodic table and are known as the f-block elements. Lanthanides are the rare earth metals, and actinides include elements like uranium (U) and thorium (Th).
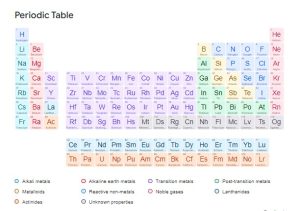
BLOCKS OF THE PERIODIC TABLE
- s-block – These are group 1 and 2.
- p-block – These are group 3 to 0/8.
- d-block – These are the elements between group 2 and 3 (i.e the transition metals).
- f-block – These are the lanthanides and actinides.
DTW Tutorials Study Resource Links;
First of All to obtain high JAMB &WAEC Scores, YOU HAVE TO Practice! Practice!! Practice!!
Use DTW JAMB & WAEC 2025 CBT Practice App!!!
– GET DTW TUTORIALS JAMB & WAEC 2025 CBT EXAM PRACTICE APP for all Subjects with over 31,000 Past Questions and Correct Solutions to Practice with offline! (Activation cost is N4000 for 1 year) Download Links Below for Mobile Phones & Laptop Computer;
DTW TUTORIALS JAMB 2025 APP For MOBILE Phone Direct Download link;
https://play.google.com/store/apps/details?id=com.iafsawii.dtw.jamb
DTW TUTORIALS JAMB 2025 APP For DESKTOP Laptop Computer Direct Download link; https://drive.google.com/file/d/1iIHBoWjEeJeCFyTO9nt-9kAveH2FqjrT/view?usp=sharing
Download Links for WAEC 2025 App;
JAMB RESOURCE LINKS BELOW;
– JAMB Past Questions Solved Playlists on Math, Phy, Chem; https://www.youtube.com/playlist?list=PLLgYU6fS5143-p4dfWIFL7keuB1SBgT2b
– THE LEKKI HEADMASTER – Summary, Questions And Answers (JAMB 2025 NOVEL); https://dtwtutorials.com/the-lekki-headmaster-jamb-2025-novel-summary-questions-and-answers-pdf-download/
– JAMB 2025 Recommended Text Books – https://dtwtutorials.com/jamb-2025-recommended-text-books-for-all-subjects/
– JAMB 2025 Syllabus all Subjects – https://dtwtutorials.com/jamb-2025-syllabus-free-download/
– JAMB 2025 Syllabus in 30 Days Timetable Challenge by DTW Tutorials for Science, Art & Commercial Subject Combinations – Cover Your JAMB Syllabus in 30 Days Challenge; https://dtwtutorials.com/jamb-2025-syllabus-in-30-days-timetable-challenge-by-dtw-tutorials-cover-your-jamb-syllabus-in-30-days-challenge/
– How to Manage Your Jamb Exam Time for High Scores; https://youtu.be/Tp4Va8haib8
– Physics Notes and Questions on All topics; https://dtwtutorials.com/category/tutorials/physics-tutorials/
– Chemistry Notes and Questions on All topics; https://dtwtutorials.com/category/tutorials/chemistry/
– How to Read, Understand and Remember Always- https://youtu.be/kL8BpRePudA
– How to Cover Your JAMB Syllabus Fast in 30 Days!!; https://youtu.be/RVgyn01Ptd0
– What to do a night before your Jamb Exam (+Exam Prayers); https://youtu.be/njbAx4Oz5Rw
– How to Manage Your Jamb Exam Time for High Scores; https://youtu.be/Tp4Va8haib8
– Overcoming Exam Fear/Anxiety– https://youtu.be/Uvf81rvd0ls
You can also join our online groups below for instant JAMB 2025 Updates;
Join DTW JAMB 2025 Intensive Tutorials Study Groups on Facebook, Telegram and WhatsApp Group;
Facebook Group – https://web.facebook.com/groups/dtwtutorialsgroup/
WhatsApp Group – https://chat.whatsapp.com/E8pprCQYtahKfpQN9UB0aU
Telegram Group – https://t.me/+AcXfhJPSIiI2ZTY0
WhatsApp Channel – https://whatsapp.com/channel/0029VaAWvTmDDmFT9o25dV3u
DTW JAMB 2025 Intensive Online Lessons/Tutorials
Online JAMB 2025 Tutorials – Your Path to Jamb Success!
Are you preparing for the JAMB 2025 Exam and aiming for excellence? Look no further than Online Jamb Tutorial by DTW Consult. We’re dedicated to helping you ace your Jamb with confidence.
- Why Choose DTW Online JAMB Intensive Tutorials?
• Engaging, Clear and Interactive Online Lectures
• Completion of JAMB Syllabus
• Weekly Quiz Assessments
• Continuous Brainstorming and Competitions
• Membership in an Active Learning Community
• Consistent Solving of JAMB Past Questions-
• Expert Jamb Instructors
• Comprehensive Study Materials - All Classes are Recorded!! In case you miss any class, and when you join us you will have access to all the previous class recorded videos!!!
• Subjects;
English
Physics
Chemistry
Biology
Math
Economics
Literature
Crs
Government
• Affordable Tuition – N7000 monthly (6pm – 10pm, Mon to Fri)
Lectures Ongoing! Register Now!!
Bank Details:
Account Name: DTW Consult
Account Number: 6414330770
Bank: Moniepoint
Amount – N7000
For easy payment and enrollment.
Proof of payment should be sent by WhatsApp.
Contact Us:
WhatsApp: 09085099582, 08038732879
Email: dtwconsultng@gmail.com
Take a step closer to your Jamb success with DTW Online Jamb 2025 Intensive Tutorials.
Let’s work together to unlock your full potential!

https://youtu.be/P7wtBH46ZMMnsive Tutorials. Let’s work together to unlock your full potential! #JambPrep #OnlineTutorial #DTWConsult #JambSuccess #jamb2025 #utme2025

No Comments